With our expansive industry knowledge and engineering expertise, Custom Powder Systems provides groundbreaking solutions in the pharmaceutical industry that redefine how companies handle complex processes,...
Read More
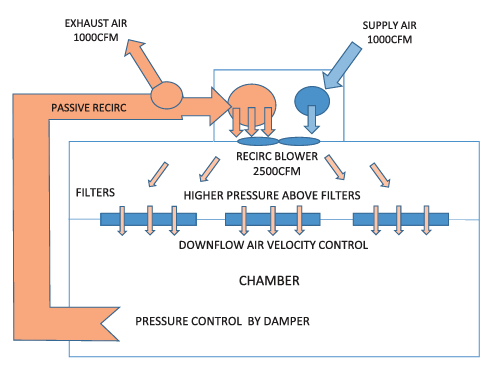
Aseptic Isolator Basic Features
The Aseptic Isolator is designed to the challenges and market requirements for low OEL containment systems in Pharmaceutical, Biotechnology, Medical devices and Radiopharmaceutical. This technology is providing a safe working environment addressing both ergonomic and safety concerns for operators in research, manufacturing, testing and handling hazardous materials. From aseptic applications, an environment capable of delivering a confident Class 100 (ISO 5) or better working environment. This equipment can be portable or stationary or grouped to other isolators to form a “production system” or complete containment solution. Isolators are designed to be tested and certified by their respective industry standards. Our experienced staff has over 50 years of containment experience. They serve on many professional societies’ boards and standards committees to keep in pace with new technology and industry requirements.

Applications
Interested in product information?
Here are download versions of product specifications and information.
Additional Information
Revolutionizing Manufacturing Efficiency: Innovative Powder Handling Solutions
Custom Powder Systems is a forerunner in the powder handling and containment sector. We pride ourselves on our extensive industry experience which allows us to...
Read MoreContainment Tool Box
Pyrophoric Material Dispensing System Enhance product safety and prevent release during transfer operations. Perfect for professionals looking for a containment tool box. The potency of...
Read More1.1 EVA SAMPLE RETRIEVAL ISOLATOR – NASA HABITAT DEMONSTRATION UNIT (HDU)
Aseptic Powder Dispensing and Blending System Project Overview The Habitat Demonstration Unit (HDU) project is a unique project from a multi-center team of NASA architects,...
Read MoreAseptic/Toxic Fill-Finish System
Aseptic/Toxic Fill-Finish System Download Project Info Project Overview This system is one of the largest automated sterile fill/finish systems in existence to date and the...
Read MoreAseptic Powder Dispensing and Blending System
Pyrophoric Material Dispensing System Download Case Study Information Fortune 100 Pharmaceutical Corporation Sterile Powder Dispense and Blending Project Project Overview – CPS prepared a system...
Read MorePyrophoric Material Dispensing System
Pyrophoric Material Dispensing System Download Project Information Project Overview CPS collaborated with sister division ICS to design, build and install this system. The system was...
Read More










